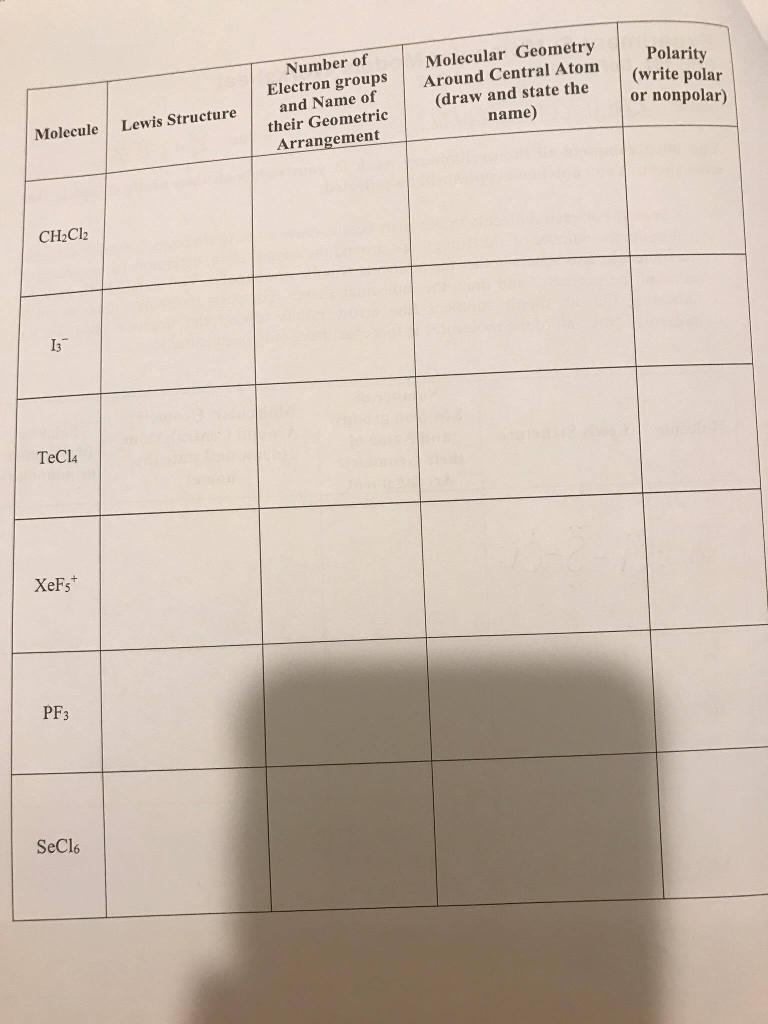
The Shorthand electron configuration (or Noble gas configuration) as well as Full electron configuration is also mentioned in the table. Understanding the electron geometry of PF3 is essential in predicting its chemical behavior and properties, including its reactivity, polarity, and interactions with other molecules.Electron configuration chart of all Elements is mentioned in the table below. This electron geometry influences the molecular geometry of PF3, which is trigonal pyramidal due to the presence of the lone pair of electrons. Therefore, the electron geometry of PF3 is tetrahedral, with the phosphorous atom at the center and the three fluorine atoms and one lone pair of electrons arranged around it. According to VSEPR theory, the lone pair of electrons will repel the bonding pairs, causing the molecule to adopt a trigonal pyramidal shape. The Lewis structure of PF3 shows that the central phosphorous atom has one lone pair of electrons and is bonded to three fluorine atoms. To determine the electron geometry of PF3, we must analyze its Lewis structure and apply the VSEPR theory. For example, the polarity of a molecule is determined by its molecular geometry, which in turn is influenced by the electron geometry. Understanding both electron and molecular geometry is crucial in predicting the chemical behavior and properties of a molecule. Select your preferences below and click Start to give it a try Number of problems: 1. Unlike electron geometry, molecular geometry takes into account the presence of lone pairs of electrons, which can affect the shape of the molecule. This online quiz is intended to give you extra practice in identifying the molecular and electron geometry of chemical compounds using VSEPR theory. In contrast, molecular geometry refers to the arrangement of atoms in a molecule, including both the central atom and surrounding atoms. This geometry is determined by the Valence Shell Electron Pair Repulsion (VSEPR) theory, which postulates that electron pairs in the valence shell of an atom repel each other, causing them to arrange themselves as far apart from each other as possible. An explanation of the molecular geometry for the HI (Hydrogen iodide) including a description of the HBr bond angles. The electron geometry of a molecule refers to the spatial arrangement of electron pairs around the central atom, including both bonding and non-bonding pairs. Maybe you are interested What Channel is HBO Max on Fios? - A Complete Guide Electron Geometry vs Molecular Geometry The physical properties of PF3, including its electron geometry, play a crucial role in determining its behavior and applications. It is also used in the production of organic compounds, as a reducing agent, and as a source of fluorine in the synthesis of other chemicals. PF3 is commonly used as a ligand in coordination chemistry, where it forms complexes with metal ions. The chemical structure of PF3 consists of one phosphorous atom bonded to three fluorine atoms, with a lone pair of electrons on the phosphorous atom. According to the VSEPR theory, if the H2S molecule ion has an AX2N2 generic formula, the molecular geometry and electron geometry will both be tetrahedral or V-bent-shaped forms. PF3 is a member of the phosphorus trifluoride family, which comprises of other compounds such as PCl3 and AsF3. The general molecular geometry formula for H2S is AX2N2. It is a colorless, toxic, and flammable gas that is soluble in organic solvents. 
PF3 is a chemical compound with the molecular formula PF3. In this article, we will discuss the electron geometry of PF3, a chemical compound that finds applications in various fields, including the production of pesticides, herbicides, and other organic compounds. Understanding the electron geometry is essential in predicting the molecular geometry, which helps to determine the polarity, reactivity, and other characteristics of the molecule. The electron geometry of a molecule refers to the arrangement of electron pairs around the central atom. Molecular geometry plays a crucial role in understanding the chemical properties and behavior of molecules. Determining the Electron Geometry of PF3.Electron Geometry vs Molecular Geometry.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
